 /PeriodicTable-56a128ab5f9b58b7d0bc938c.jpg)
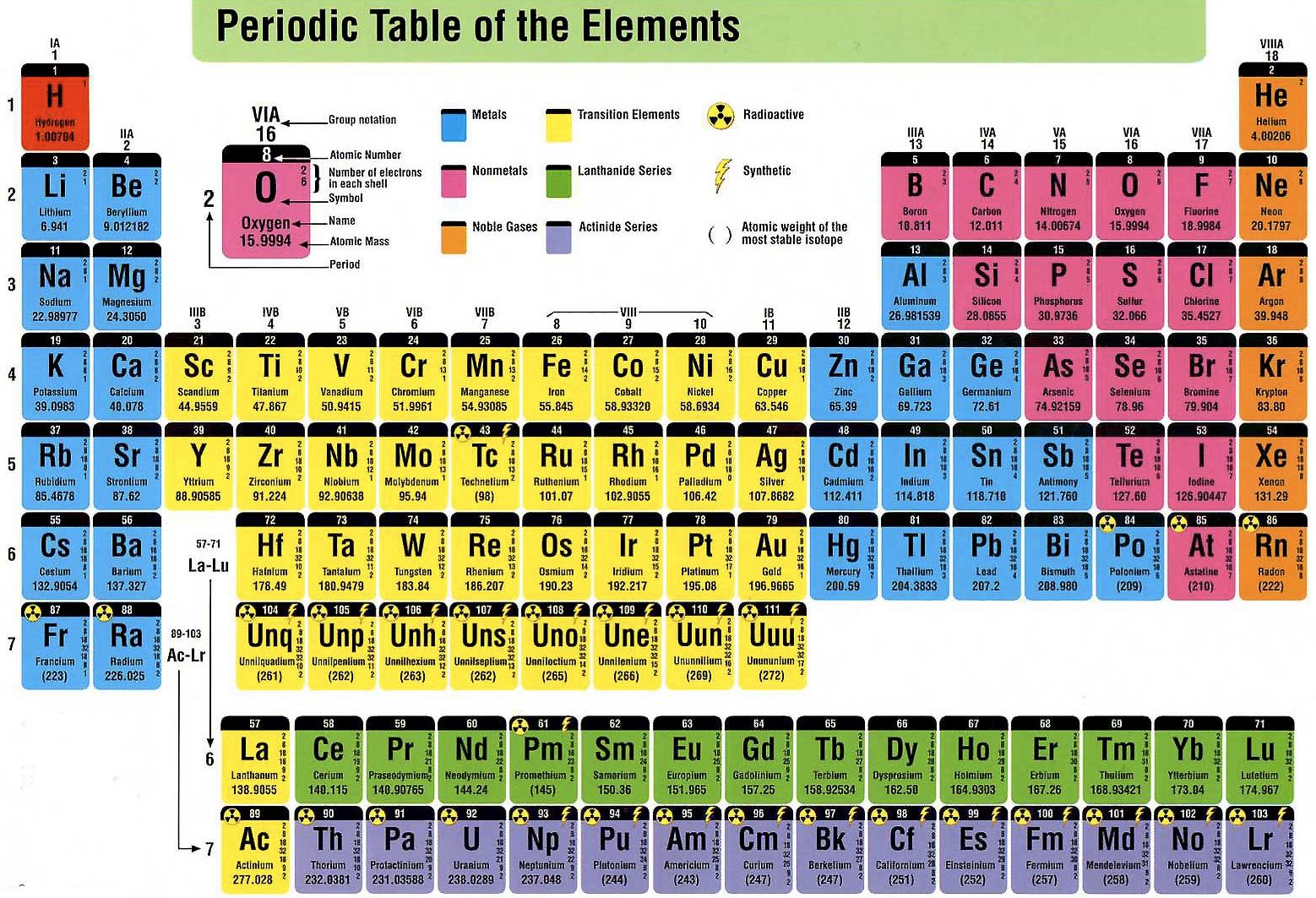
Another example is the alkali metals which all align on the left-most column. They all have a full outer shell of electrons, making them very stable (they tend not to react with other elements). These elements all line up in the eighteenth or last column of the periodic table. One example of a group is the noble or inert gases. There are 18 columns or groups and different groups have different properties. Groups are the columns of the periodic table. In each period the left most element has 1 electron in its outer shell and the right most element has a full shell. The first one is short and only has two elements, hydrogen and helium. There are seven (or eight) total periods. When they are lined up this way, elements in the columns have similar properties.Įach horizontal row in the table is a period. Some columns are skipped in order for elements with the same number of valence electrons to line up on the same columns. From left to right elements are lined up in rows based on their atomic number (the number of protons in their nucleus). ISBN 978-0-19-530573-9.It is called "periodic" because elements are lined up in cycles or periods. The periodic table, its story and its significance. Period 7: Fr, Ra, Ac, Th, Pa, U, Np, Pu, Am, Cm, Bk, Cf, Es, Fm, Md, No, Lr, Rd, Db, Sg, Bh, Hs, Mt, Ds, Rg, Cn, Nh, Fl, Mc, Lv, Ts, Og (all elements are radioactive contains heaviest natural elements and many synthesized elements).Period 6: Cs, Ba, La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu, Hf, Ta, W, Re, Os, Ir, Pt, Au, Hg, Tl, Pb, Bi, Po, At, Rn (first period with f-block elements).Period 5: Rb, Sr, Y, Zr, Nb, Mo, Tc, Ru, Rh, Pd, Ag, Cd, In, Sn, Sn, Te, I, Xe (same number of elements as period 4, same general structure, and includes the first exclusively radioactive element, Tc).Period 4: K, Ca, Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Ga, Ge, As, Se, Br, Kr (first period with d-block elements).Period 3: Na, Mg, Al, Si, P, S, Cl, Ar (all have at least 1 stable isotope).Period 2: Li, Be, B, C, N, O, F, Ne (involves s and p orbitals).Period 1: H, He (does not follow the octet rule).Note that the lanthanides are within period 6 and the actinides are in period 7. Some periods contain more elements than others because the number of included elements depends on the number of electrons allowed in an energy sublevel. Elements within a period display periodic table trends, moving from left to right, involving atomic and ionic radius, electronegativity, There are seven element periods. Element PeriodsĮlements within a period share the same number of electron shells and the same highest unexcited electron energy level. The alkali metals, alkaline earths, lanthanides, actinides, transition metals, and basic metals are all groups of metals. The metalloids have properties intermediate between metals and nonmetals. The nonmetals, halogens, and noble gases are all types of nonmetals. Under this system, hydrogen is a nonmetal. These groups go by the names alkali metals, alkaline earth metals, transition metals, basic metals, nonmetals, halogens, noble gases, lanthanides, and actinides. Sometimes chemists classify element groups according to shared properties, which do not strictly adhere to individual columns. The group 17 elements are highly reactive, colorful nonmetals. For example, the group 1 elements are all soft, reactive metals. Elements in the same group share common chemical and physical properties. The lanthanides and actinides, located below the main table, all fit within group 3. For example, elements in group 1 have 1 valence electron, elements in groups 3-12 have a variable number of valence electrons, and elements in group 17 have 7 valence electrons. The number of valence electrons depends on the octet rule. Atoms in a period have the same number of electron shells.Įlements within the same group share the same number of valence electrons. An element period is a horizontal row on the periodic table.Atoms in a group share the same number of valence electrons. An element group is a vertical column on the periodic table.Element atomic number increases as you move down a group from top to bottom or across a period from left to right. Both groups and periods reflect the organization of electrons in atoms. A group is a vertical column down the periodic table, while a period is a horizontal row across the table. Groups and periods organize elements on the periodic table of the elements. This entry was posted on Jby Anne Helmenstine (updated on June 28, 2021)Ī periodic table group is a column, while a periodic table period is a row.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
